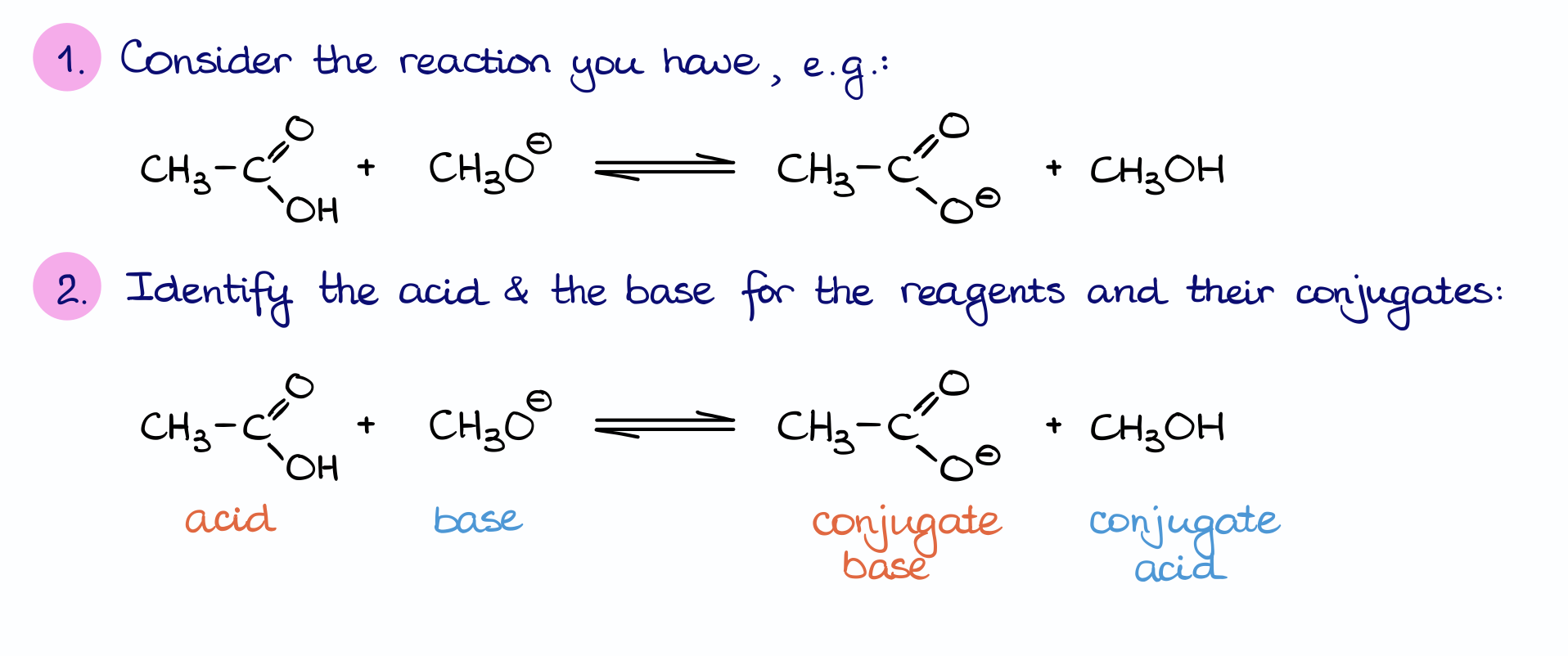
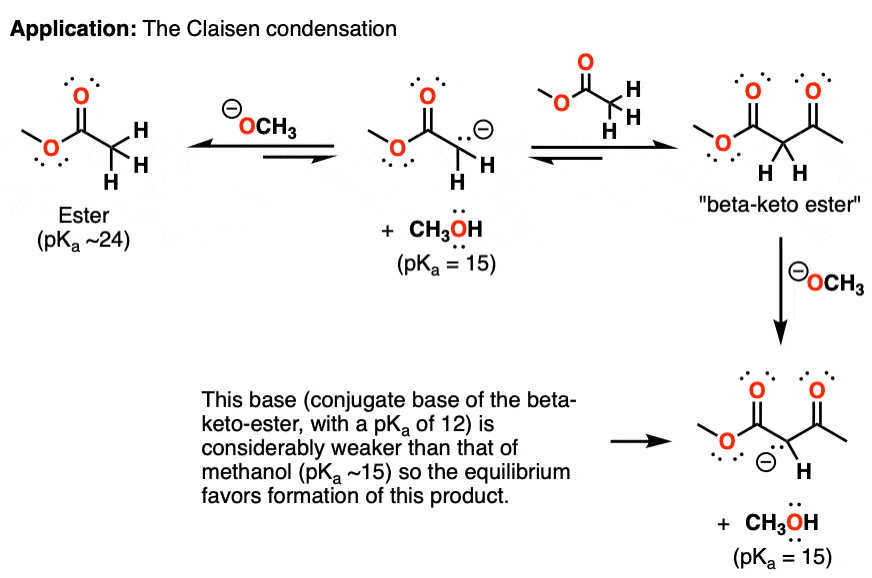
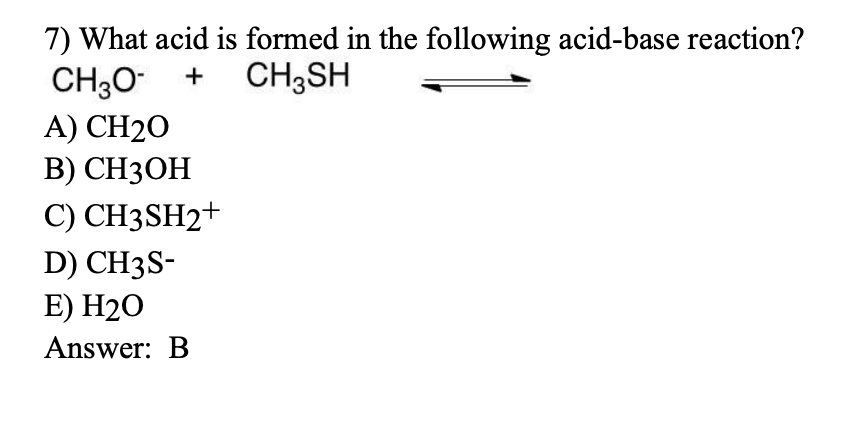
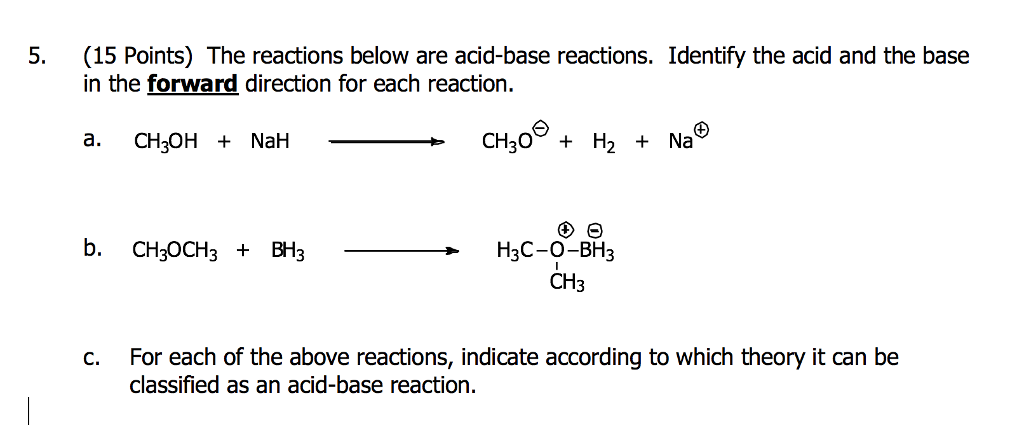
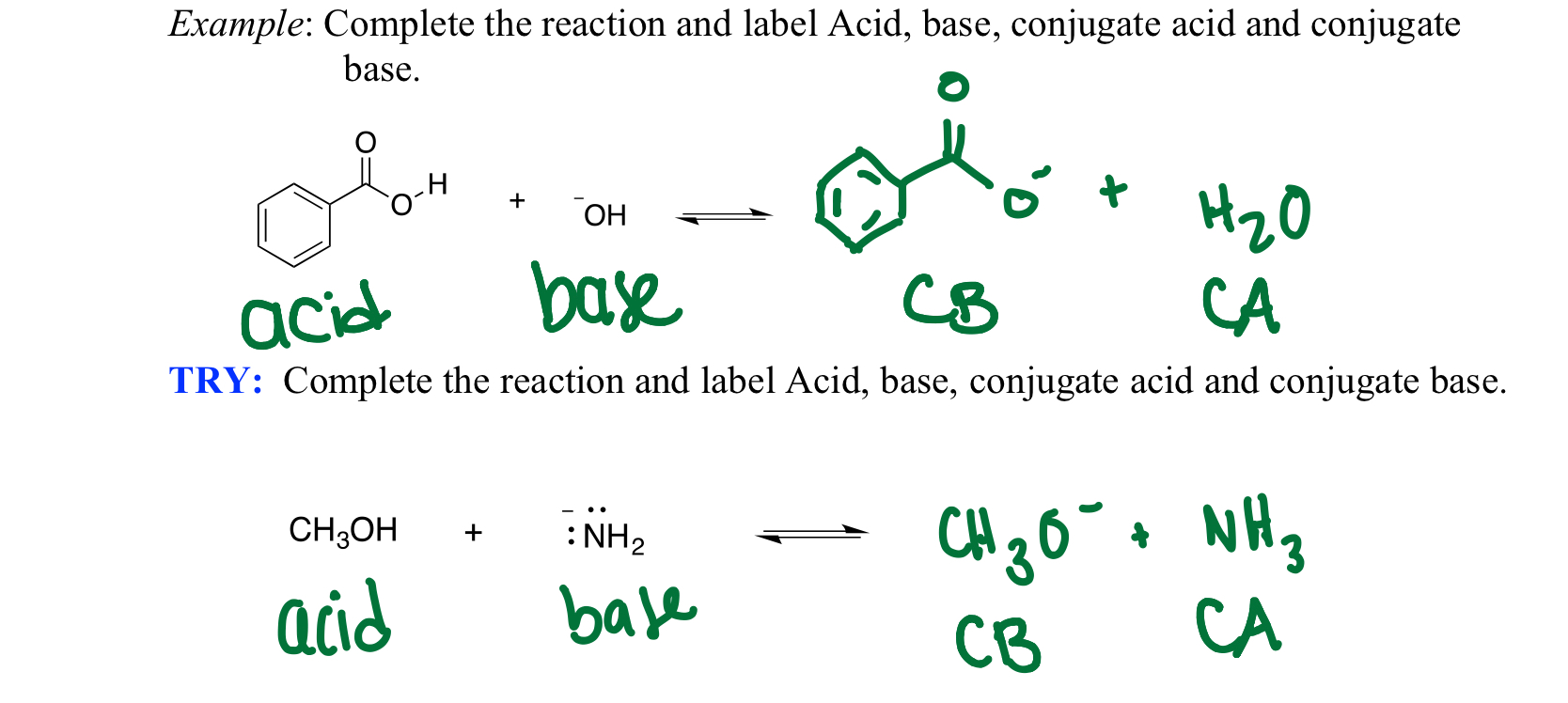
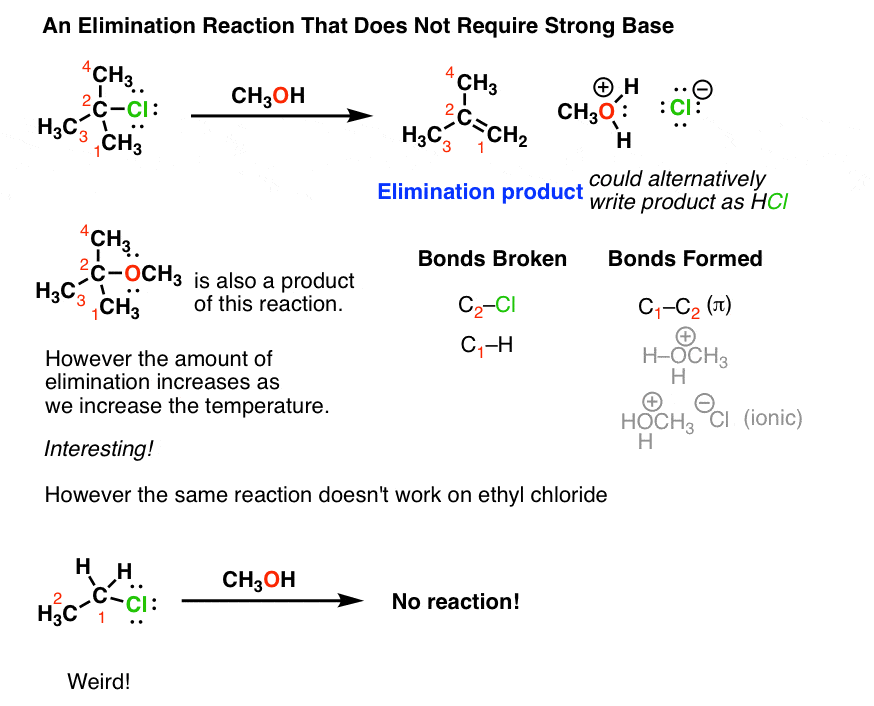
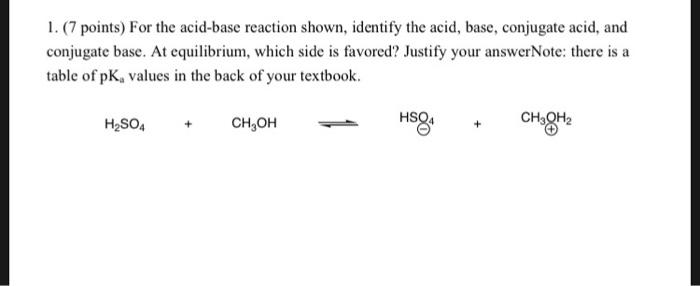
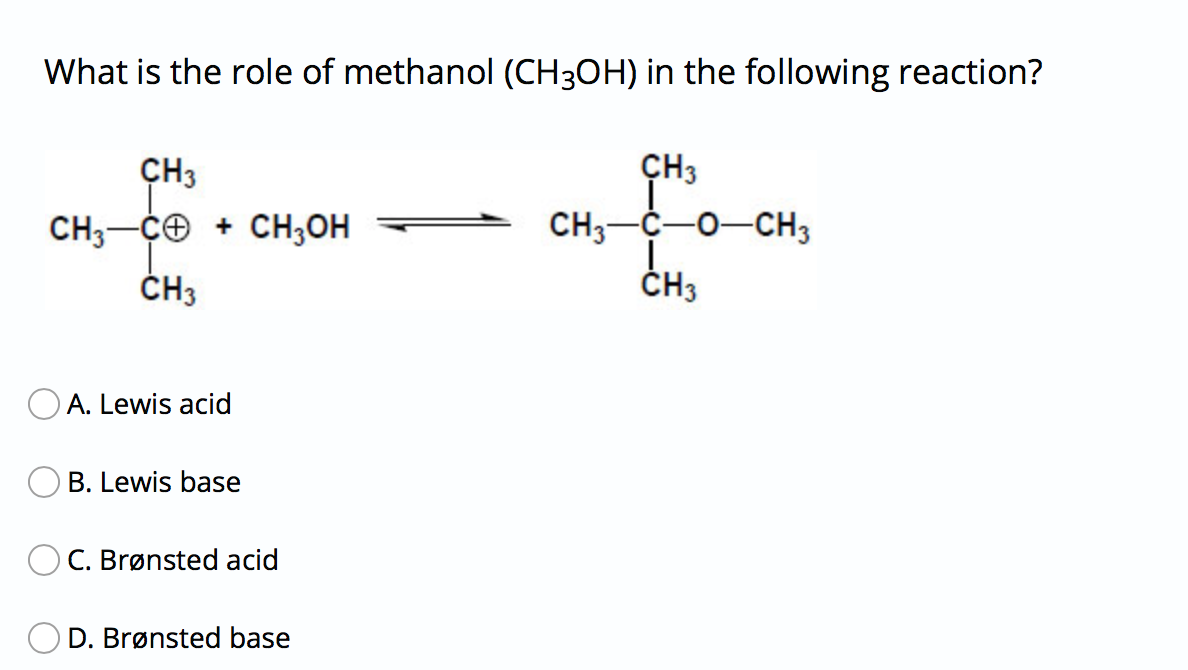
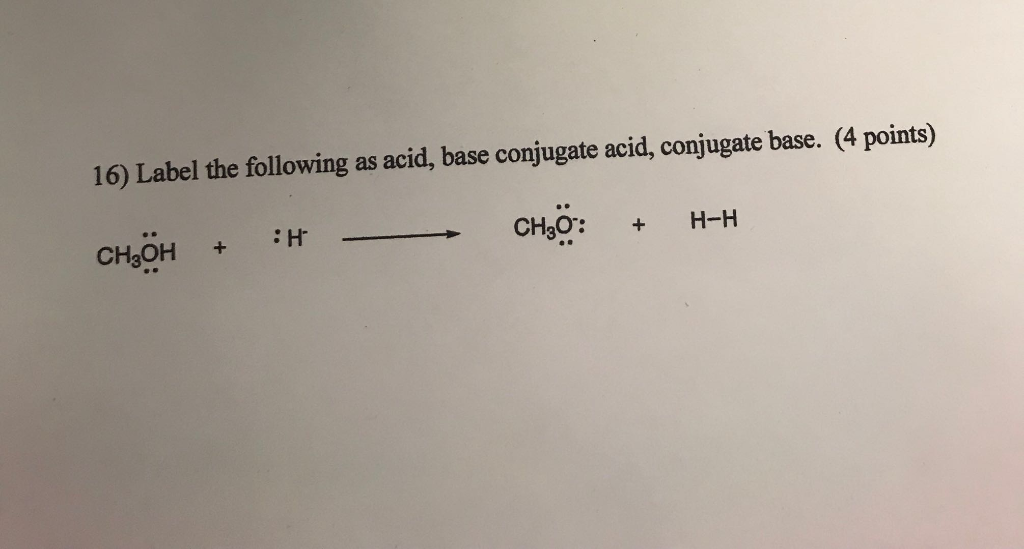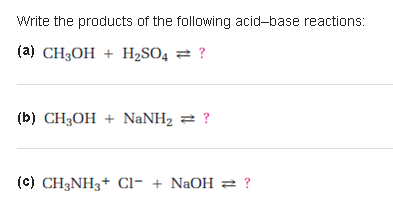Ch2 Acid-Base Part2-1 - Ch2 Acid-Base Part2-1 - 1 What conjugated base is formed in the following - Studocu
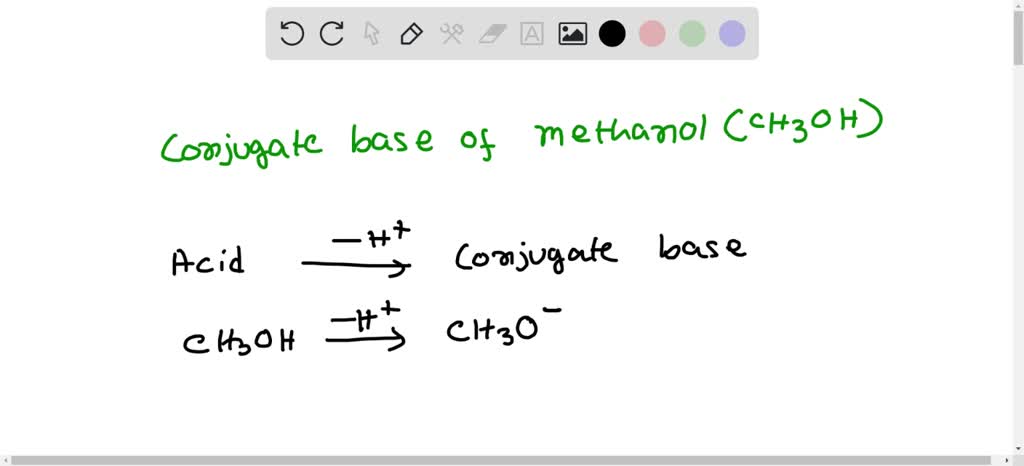
SOLVED: '17) When methanol (CHzOH) acts as a base, its conjugate acid is A) -CH2OH B) CH3O- C) CHAOH D) CHzOH2+ E) CH4Ot'
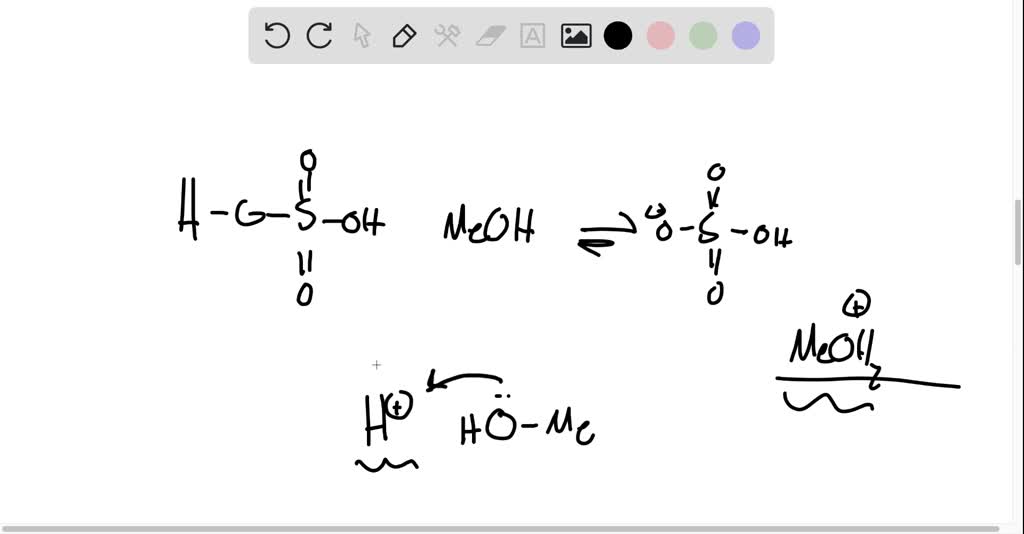
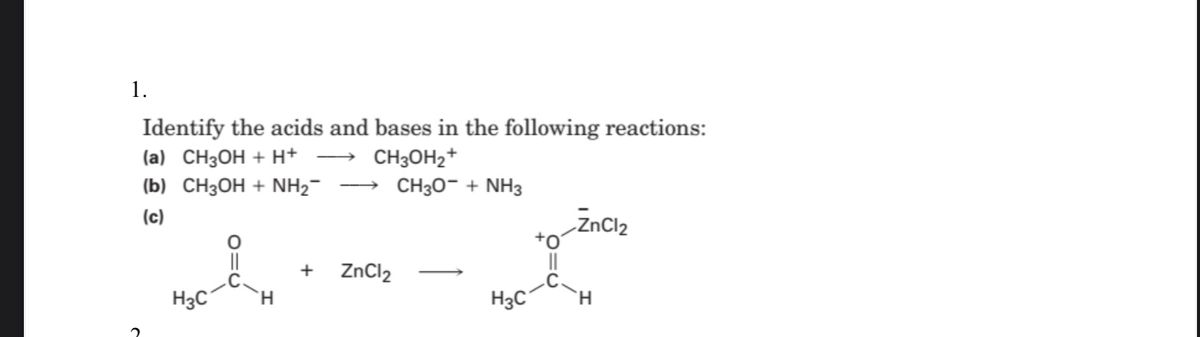
SOLVED: Organic Chemistry Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 <–> ? (b) CH3OH + NaNH2 <–> ? (c) CH3NH3 + Cl- + NaOH <–> ? -
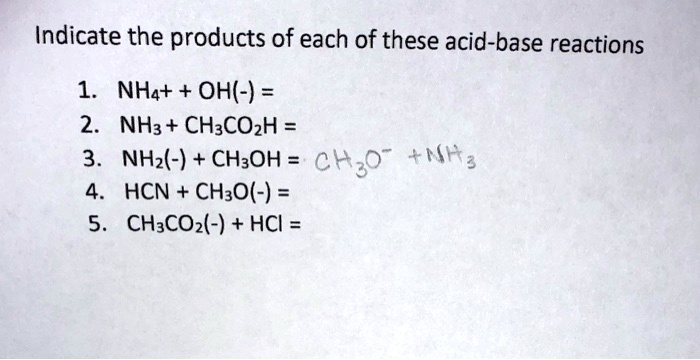
SOLVED: Indicate the products of each of these acid-base reactions 1. NHa+ + OH(-) = 2. NHz + CH3COzH = 3. NHz(-) + CH3OH = chzo +Mtg HCN + CHzO(-) = 5 CH:COz(-) + HCI =