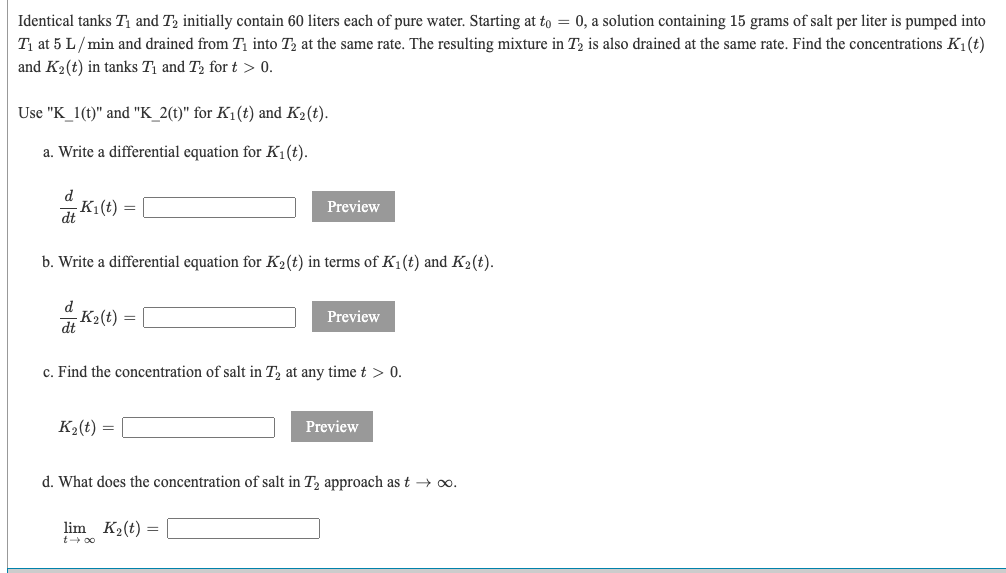
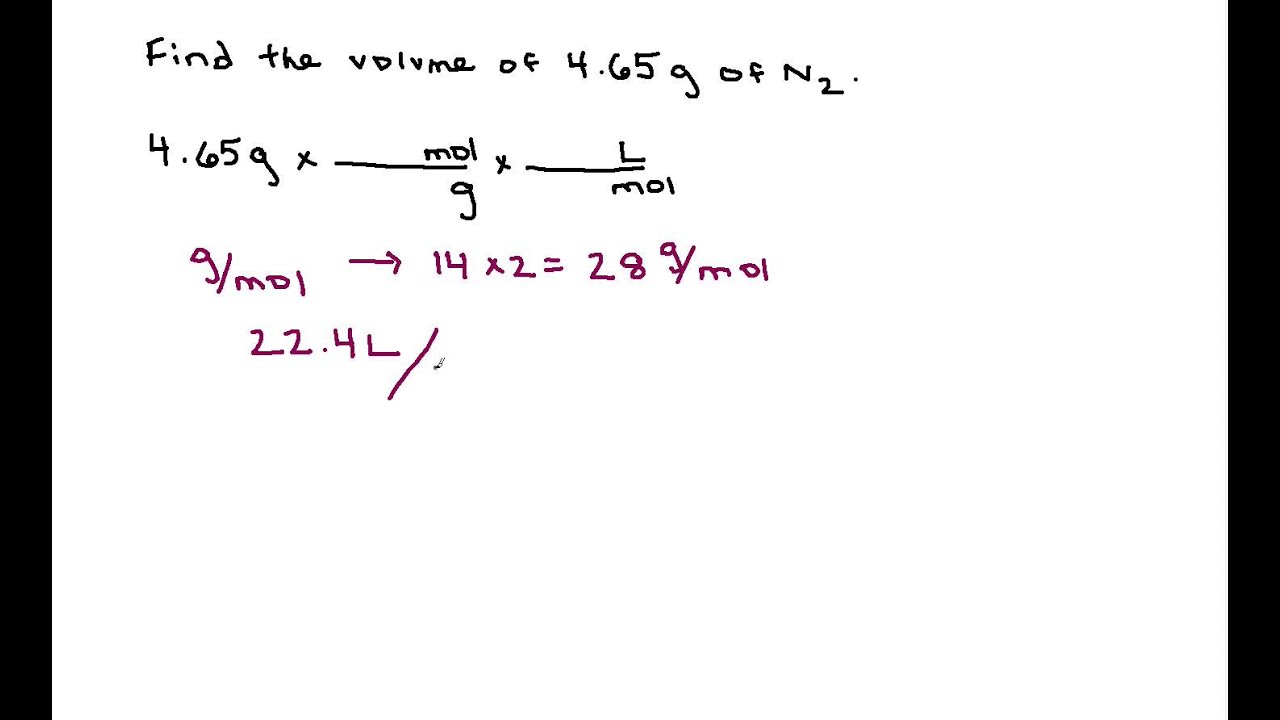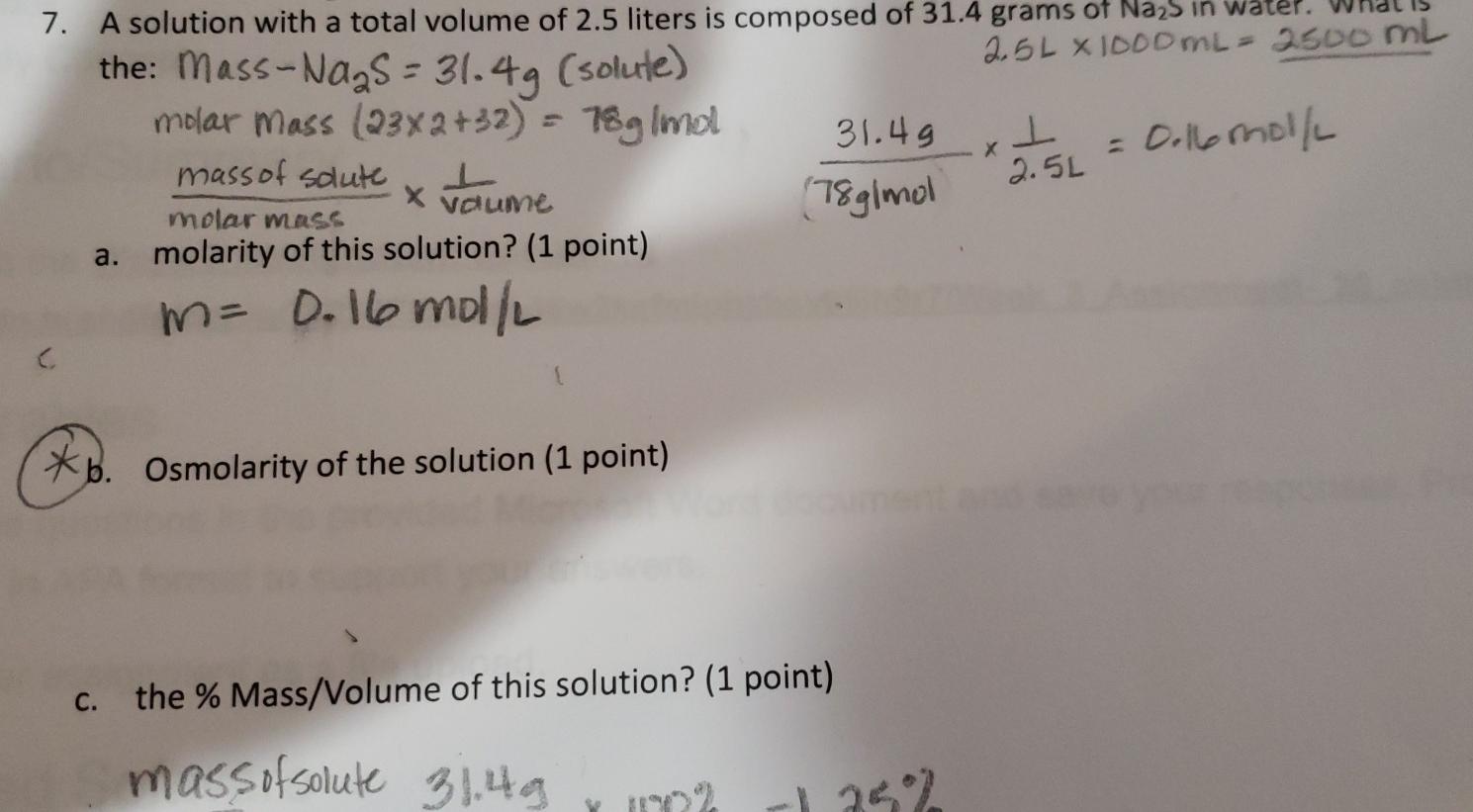SOLVED: points) Initially 15 grams of salt are dissolved into 10 liters of water: Brine with concentration of salt 3 grams per liter is added at a rate of 4 liters per

Molarity Dilution Problems Solution Stoichiometry Grams, Moles, Liters Volume Calculations Chemistry - YouTube
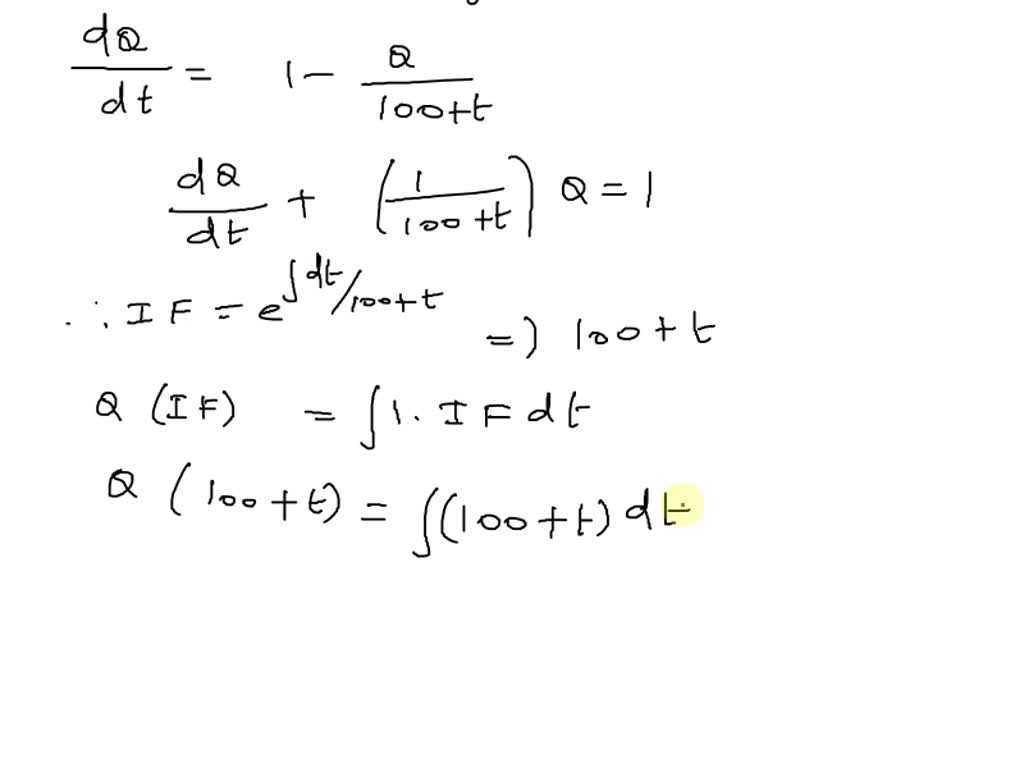
SOLVED: '(25pts) A 500-liter tank initially contains 10 gram of salt dissolved in 200 liters of water. Starting to 0, water that contains 1/4 gram of salt per liter is poured into

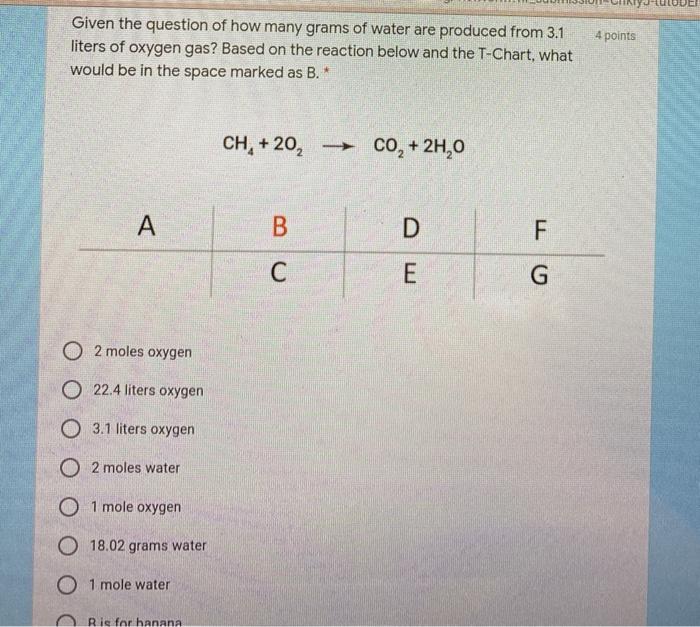
SOLVED: How many liters of oxygen gas are needed to produce 110.0 grams of water at 25.0 C and 1.00 atm pressure according to the chemical equation 2 H2(g) + O2(g) —> 2 H2O(l)?

Molarity Molarity = grams / molar mass / Liters Liters = grams / molar mass/ Molarity Grams = Molarity x Liters x molar mass. - ppt download