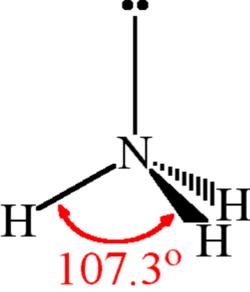
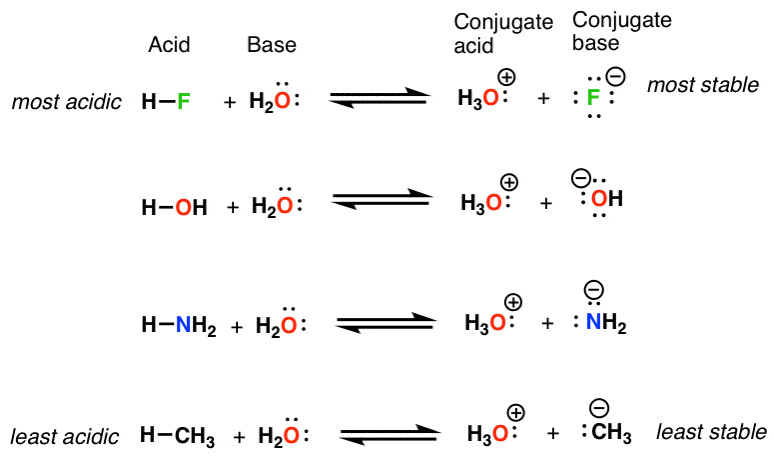
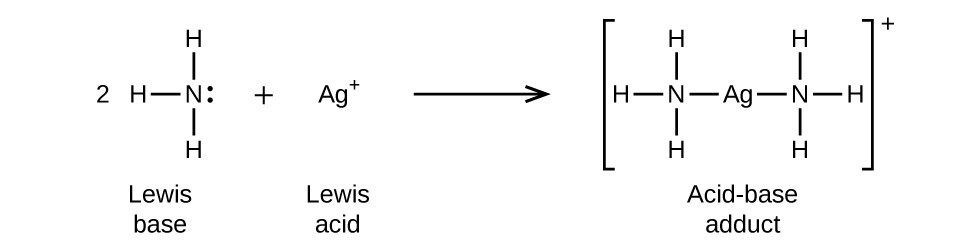
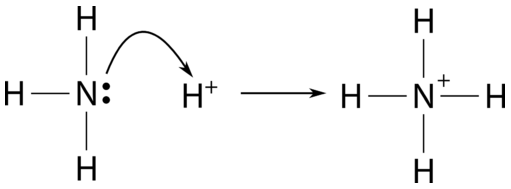
NH3 is a weak base (Kb = 1.8 times 10^-5) and so the salt NH4Cl acts as a weak acid. What is the pH of a solution that is 0.050 M in

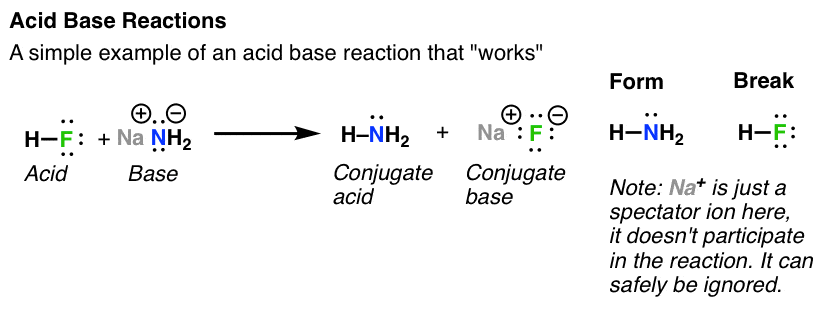
SOLVED: Identify the conjugate acid and base pairs in the following acid base reaction: HCO3 NH3 CO3-2 NH4t Acidlconjugate base: HCO3 CO3-2 Baselconjugate acid NH3 NH4 B Acidlconjugate base: CO3-2 / HCO3"

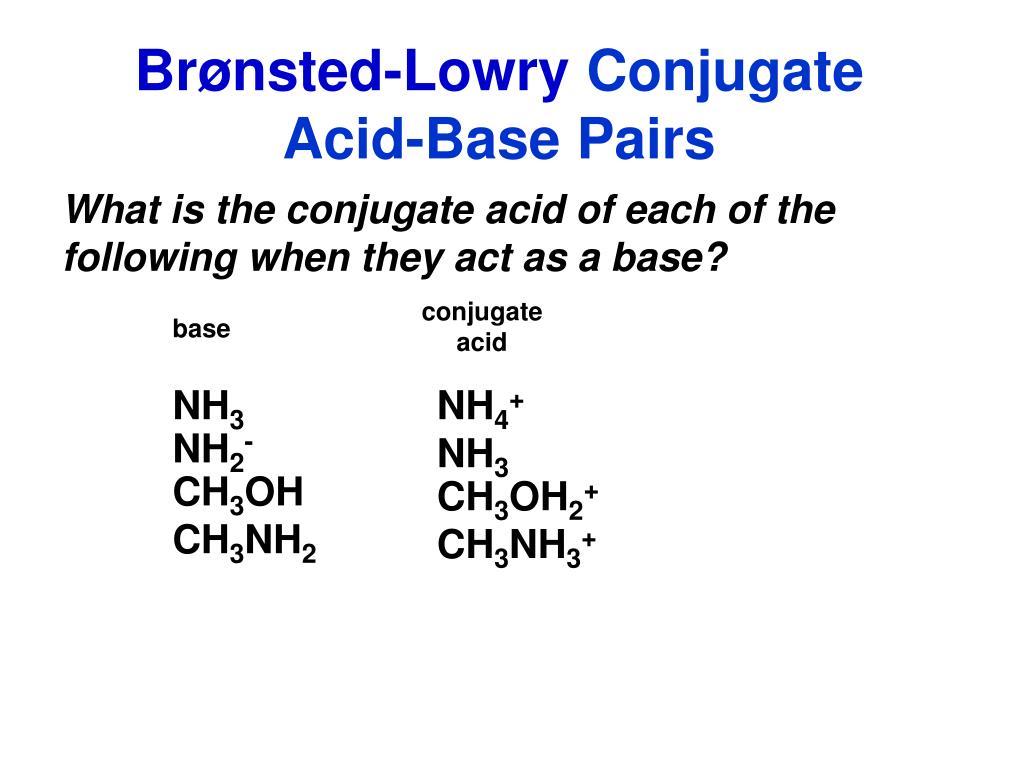
Identify the conjugate acid-base pairs in this equilibrium. NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com

In the following acid-base reaction, HPO42- is the H2PO4- (aq) + NH3(aq) → HPO42- (aq) + - Brainly.com