![Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube](https://i.ytimg.com/vi/SLPu7qlUdEA/maxresdefault.jpg)
Henderson Hasselbalch Equation Acid Base Buffer Chemistry Introduction ph = pka + log [A/HA] - YouTube

BT_GS 1.9 Describe factors influencing the distribution of drugs (for example …. pH, pKa) …. | Primary LO of the Day

pH, pOH, H3O+, OH-, Kw, Ka, Kb, pKa, and pKb Basic Calculations -Acids and Bases Chemistry Problems - YouTube
![SOLVED: Salt of a Strong Acid and a Weak Base pKa log [S] pH Salt of a Strong Base and a Weak Acid pKa pKw Jog [S] pH Salt of a Weak SOLVED: Salt of a Strong Acid and a Weak Base pKa log [S] pH Salt of a Strong Base and a Weak Acid pKa pKw Jog [S] pH Salt of a Weak](https://cdn.numerade.com/ask_images/21c70eb322cb4658bb80b94de9a1c863.jpg)
SOLVED: Salt of a Strong Acid and a Weak Base pKa log [S] pH Salt of a Strong Base and a Weak Acid pKa pKw Jog [S] pH Salt of a Weak
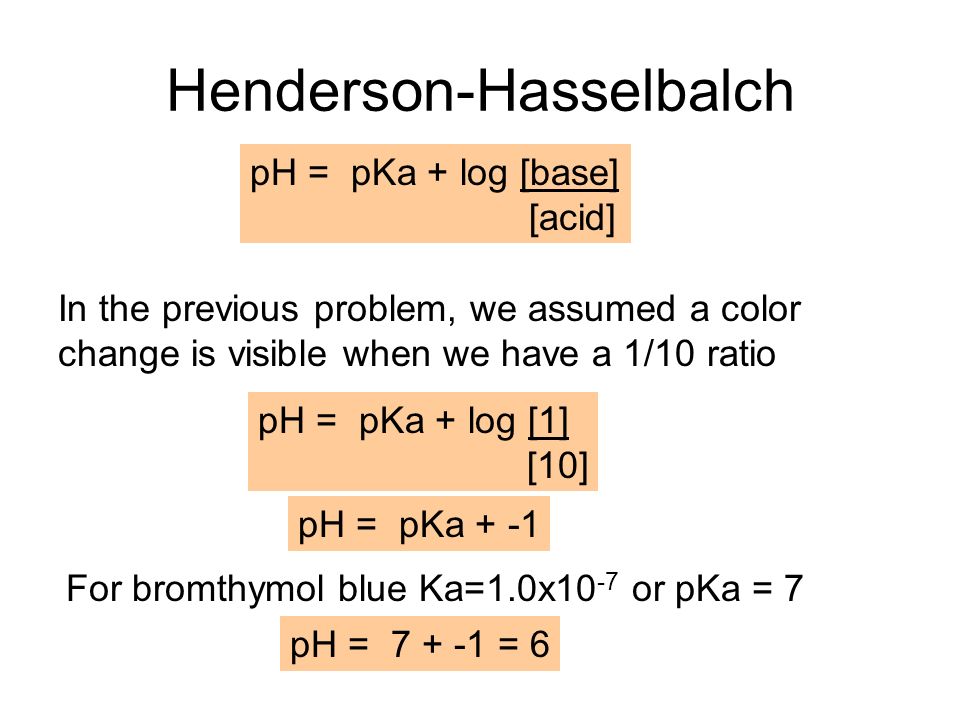
Acid Base Titrations AP Chemistry Chapter 15. Titration Titrations are used to determine the amount of acid or base in a solution Titrant: the solution. - ppt download

Yexel Exams - Buffer solutions are used to reduce pH fluctuations associated with introduction of small amounts of strong acids or bases. Typical buffer solutions are composed of a weak acid or
![SOLVED: Henderson-Hasselbalch Equation (YOu must be able to derive this equation): [conjugate base] [A-] pH pKa + log OR pH = pKa + log [acid] [HA] 0. 1. What is the pH SOLVED: Henderson-Hasselbalch Equation (YOu must be able to derive this equation): [conjugate base] [A-] pH pKa + log OR pH = pKa + log [acid] [HA] 0. 1. What is the pH](https://cdn.numerade.com/ask_images/b0b5d45010ed4627b06a3a093e4b025c.jpg)


![Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube](https://i.ytimg.com/vi/2jpB30LsT8g/maxresdefault.jpg)


![Solved Important equations pH = pKa + log[salt]/[acid] | Chegg.com Solved Important equations pH = pKa + log[salt]/[acid] | Chegg.com](https://media.cheggcdn.com/media/14b/14b35f43-4468-42d2-a7e2-d924b3fd6eba/phpQPtBXO.png)

:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)

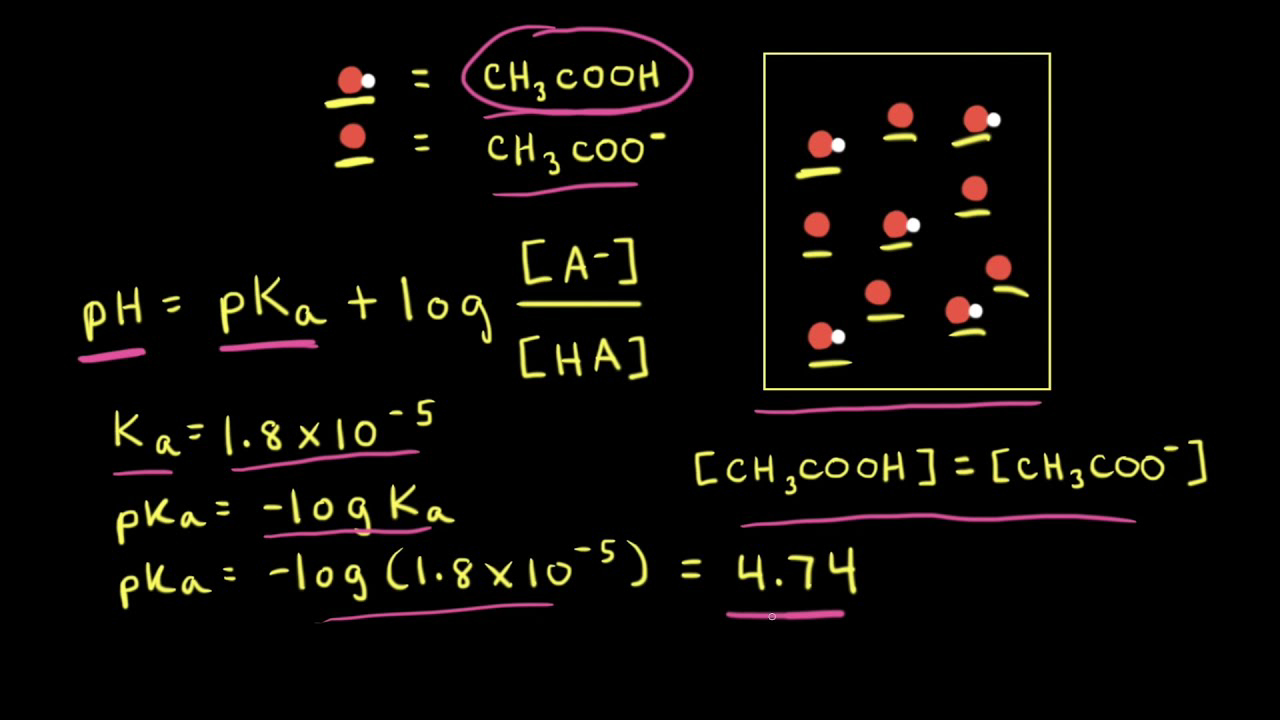





![Solved Just as pH is the negative logarithm of (H30+], pK, | Chegg.com Solved Just as pH is the negative logarithm of (H30+], pK, | Chegg.com](https://media.cheggcdn.com/media/375/3755951c-9b2e-4931-a914-33bb100113d8/php72x3Cn.png)

