![DABCO‐Mediated [4+1] Cycloaddition of β,β‐Dihalo Peroxides with Sodium Azide toward Isoxazoles - Ma - 2020 - Asian Journal of Organic Chemistry - Wiley Online Library DABCO‐Mediated [4+1] Cycloaddition of β,β‐Dihalo Peroxides with Sodium Azide toward Isoxazoles - Ma - 2020 - Asian Journal of Organic Chemistry - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/bc0b6029-aa99-47ae-9c2f-1ab222f1c0b6/ajoc202000153-fig-5001-m.jpg)
DABCO‐Mediated [4+1] Cycloaddition of β,β‐Dihalo Peroxides with Sodium Azide toward Isoxazoles - Ma - 2020 - Asian Journal of Organic Chemistry - Wiley Online Library
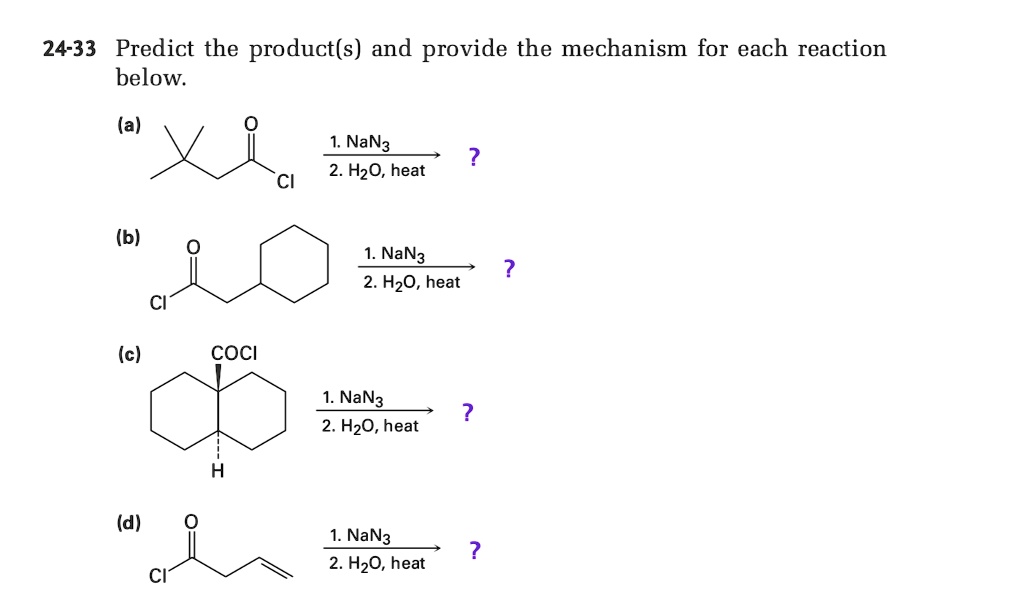
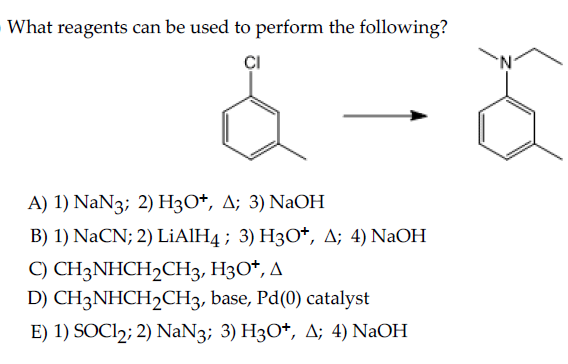
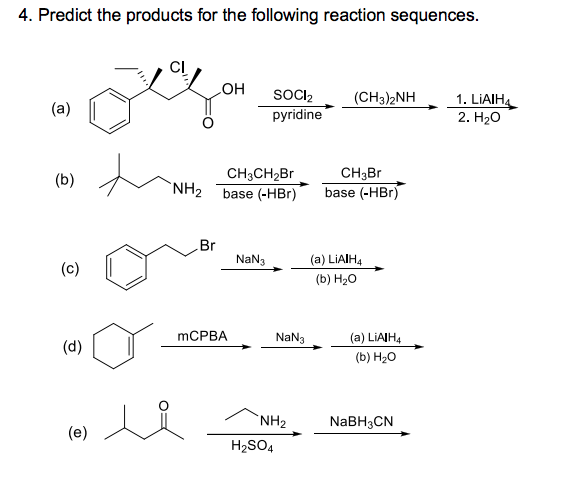
SOLVED: 24-33 Predict the product(s) and provide the mechanism for each reaction below: (a) NaN3 H2O, heat (b) NaN3 2. HzO, heat (c) coci NaN3 2. HzO, heat (d) NaN3 2. HzO, heat

Question Video: Determining the Percentage of the Wasted Starting Materials Used to Produce Sodium Azide (NaN_3) Using Atom Economy Calculations | Nagwa

Reaction of Allenyl Esters with Sodium Azide: An Efficient Synthesis of E-Vinyl Azides and Polysubstituted Pyrroles | The Journal of Organic Chemistry
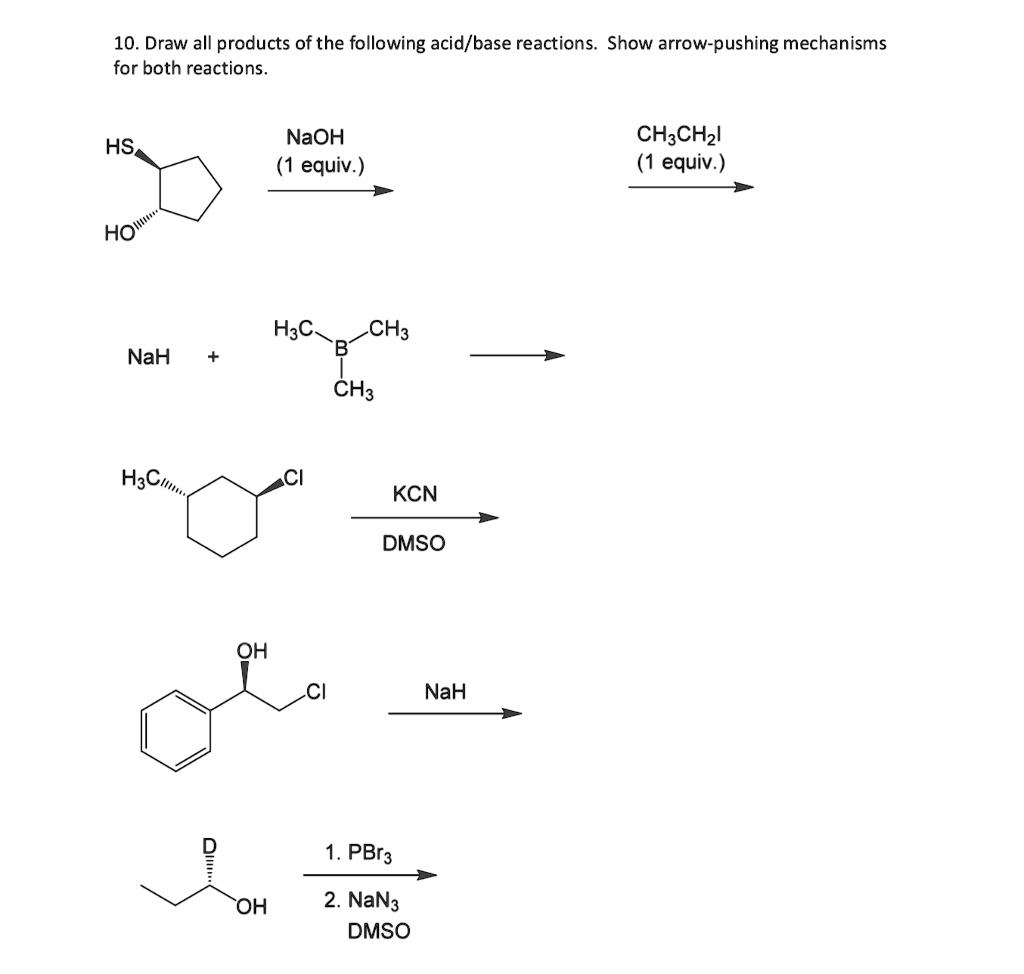
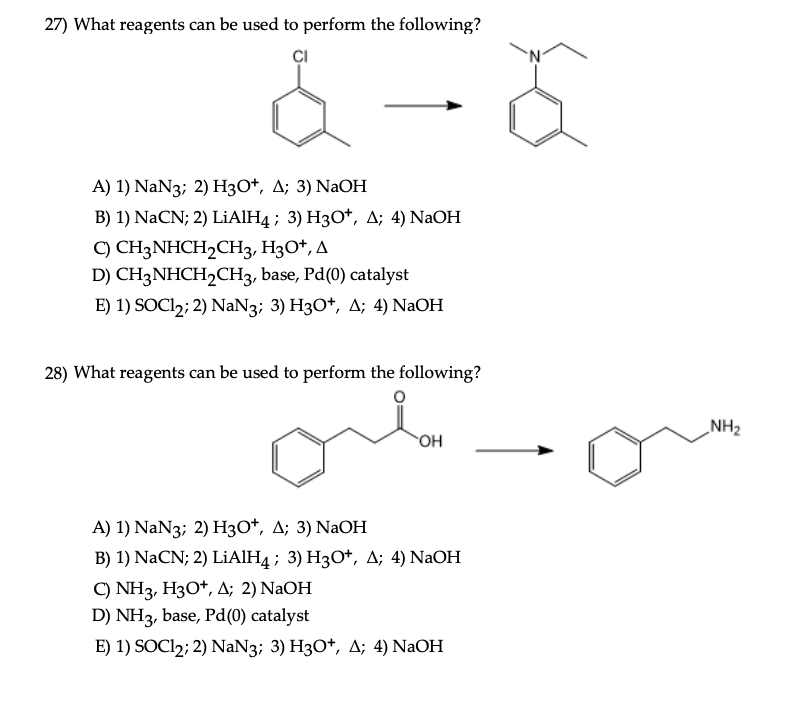
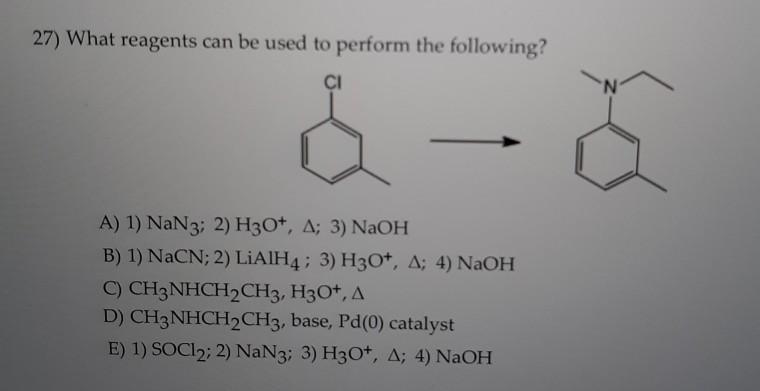
SOLVED: 10. Draw all products of the following acid/base reactions Show arrow-pushing mechanisms for both reactions. HS NaOH equiv.) CH3CHzl equiv.) H3C CH3 NaH CH3 HyCum KCN DMSO QH NaH PBr3 OH

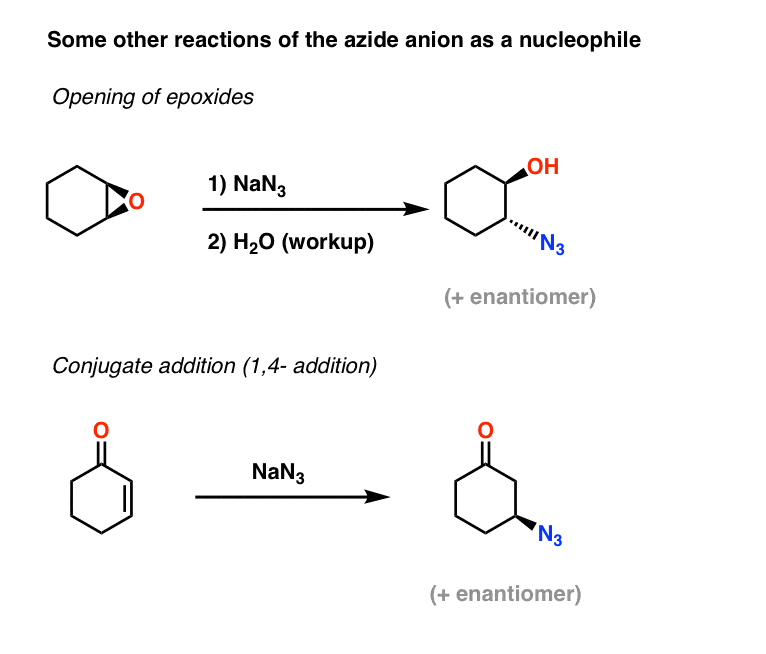
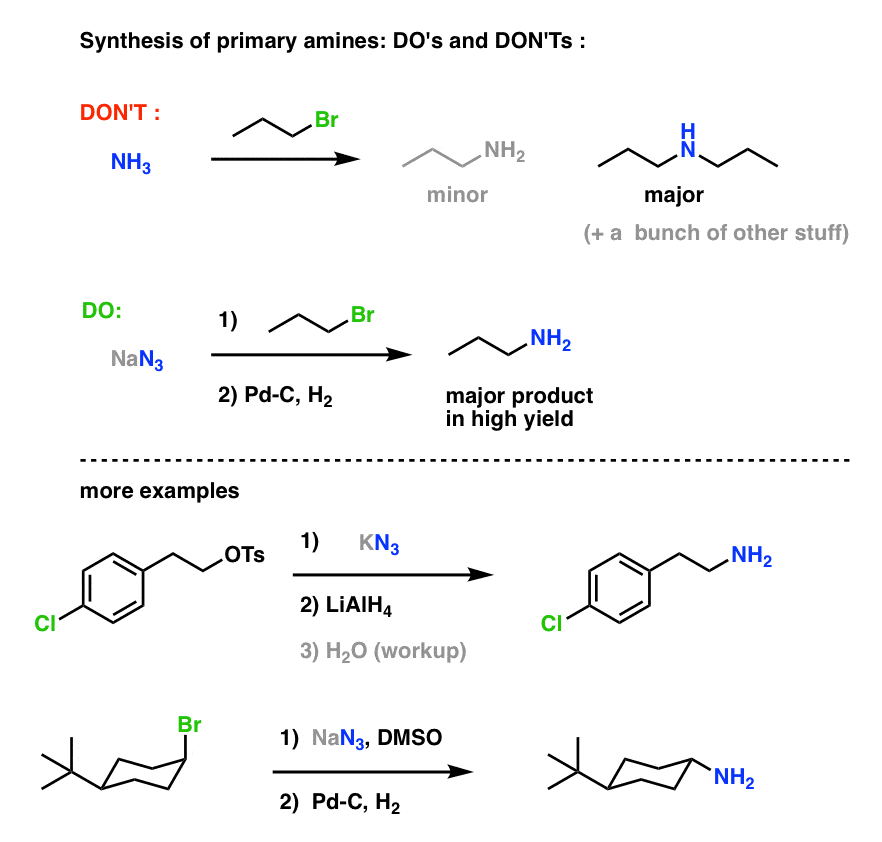
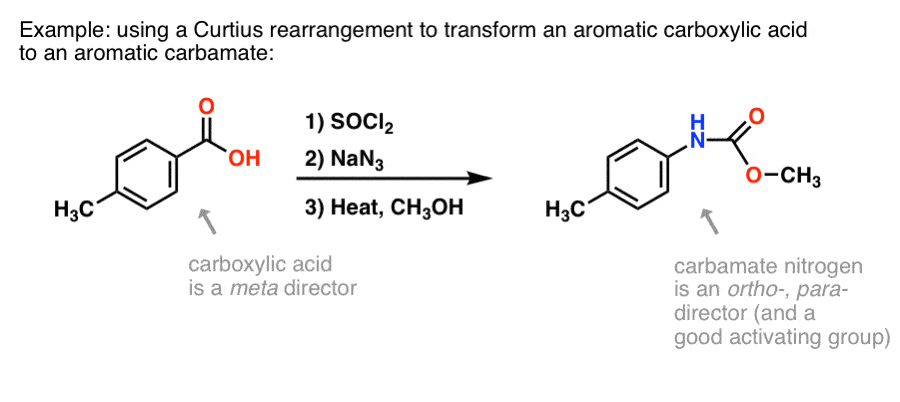
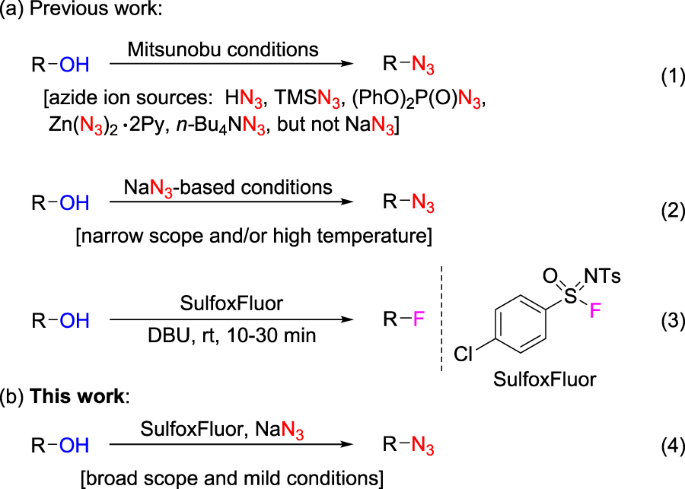
Highly regioselective ring opening of epoxides using NaN3: a short and efficient synthesis of (−)-cytoxazone - ScienceDirect

Synthetic route for ligands L1-L4, reaction conditions (i) NaN3, LiCl,... | Download Scientific Diagram
![PDF] Mutagenic effect of sodium azide (NaN3) on M2 generation of Brassica napus L. (variety Dunkled) | Semantic Scholar PDF] Mutagenic effect of sodium azide (NaN3) on M2 generation of Brassica napus L. (variety Dunkled) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8b0433bb0c78f38c2279af2abb9ebb7099521a5d/3-Figure1-1.png)
PDF] Mutagenic effect of sodium azide (NaN3) on M2 generation of Brassica napus L. (variety Dunkled) | Semantic Scholar

Air‐Induced One‐Pot Synthesis of N‐Sulfonylformamidines from Sulfonyl Chlorides, NaN3, and Tertiary/Secondary Amines - Bi - 2019 - European Journal of Organic Chemistry - Wiley Online Library