Mutations in Asn 125 of MYB.Ph3 influence base preferences at position... | Download Scientific Diagram

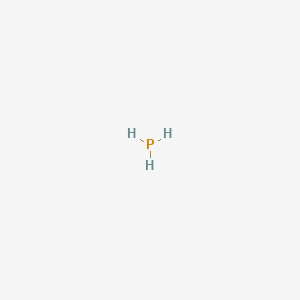
PH3 Lewis Structure (Phosphine) | PH3 Lewis Structure (Phosphine) Welcome back to Geometry of Molecules where we make Chemistry fun and easy. For today's video we are going to share the...

Draw the Lewis structure for PH3. How many bonds and non-bonding pairs are around the central atom, and what is the shape of this molecule? | Homework.Study.com

In which of the following arrangements, the sequence is not strictly according to the property written against it?

Taming PH3: State of the Art and Future Directions in Synthesis | Journal of the American Chemical Society
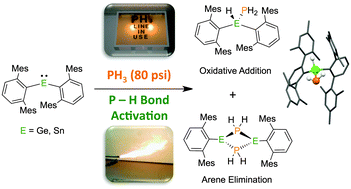
Activation of gaseous PH3 with low coordinate diaryltetrylene compounds - Chemical Communications (RSC Publishing)